Additional equipment:
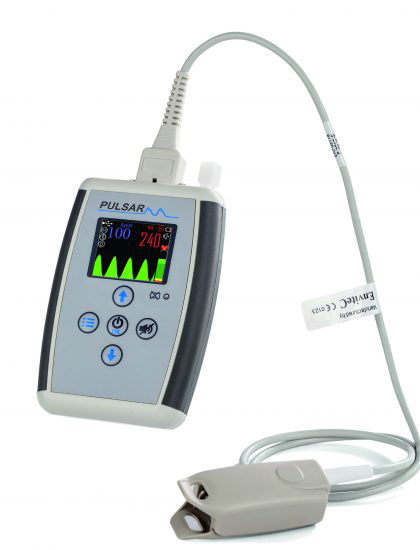
Portable pulse oximeter Pulsar-M2
Pulse oximeter Pulsar-M2
Pulsar portable pulse oximeters are diagnostic medical devices for measuring the level of saturation with oxygen in capillary blood (SpO2) and pulse rate. Pulse oximeters are non-invasive measuring instruments that are simple and easy to use. The principle of a pulse oximeter is based on transillumination of tissues with light beams with separation at red and infrared wavelengths, pulse wave digitization and signal quality control.
Pulsar-M2 pulse oximeter is apnea monitor with 24-hour recording of photoplethysmogram, spirogram, body position indicator with data storage in the built-in memory and their subsequent transfer and analysis to PC. The device records the state of apnea/hypopnea with determination of indices of apnea/hypopnea and desaturation. The device is certified.
The following protocols are available:
- 24-hour monitoring;
- sleep monitoring;
- stress.
Monitoring protocols provide statistical reports characterizing oxygen status of organism with time spent by patient in various zones of saturation and pulse rate, apnea/hypapnea index, desaturation index and other indicators with a clear graphical presentation, providing full screening “at a glance”. Long-term monitoring of SPO2 and pulse rate makes it possible to objectively confirm hypoxia, identify conditions and time of its occurrence, and reasonably select groups of patients for subsequent referral to cardiorespiratory monitoring and/or polysomnographic research.
Stress protocol of the device provides control of effect of 6-minute step test, standardized bicycle ergometric exercise, procedures in a pressure chamber, “Mountain air” procedure, allows for assessing the patient’s physical capabilities, and making a conclusion about the degree of respiratory failure in accordance with the instructions of the Ministry of Health of the Republic of Belarus “Method for Determining the Severity of Chronic Respiratory Failure in Patients with Chronic Obstructive Pulmonary Disease”.
Methodical literature
Use Instruction of the Ministry of Health of the Republic of Belarus “Method for determining severity of chronic respiratory failure in patients with chronic obstructive pulmonary disease”
Authors: Candidate of Medical Sciences D.V. Lapitsky, Doctor of Medical Sciences, Professor N.P. Mitkovskaya, Candidate of Medical Sciences E.I. Davidovskaya, A.N. Ryapolov, V.A. Pupkevich, T.V. Chirikova, R.F. Ermolkevich.
Methodical recommendations “Diagnostic capabilities of non-Invasive monitoring of arterial blood hemoglobin saturation with oxygen in clinic of internal diseases.”
Authors: Candidate of Medical Sciences D.V. Lapitsky; R.F. Ermolkevich; S.M. Metelsky; A.N. Ryapolov; Doctor of Medical Sciences, prof. N.P. Mitkovskaya; Candidate of Physical and Mathematical Sciences I.A. Manichev; Candidate of Physical and Mathematical Sciences V.G. Shcherbitsky. – Minsk: BSMU, 2017.– 68 p.
| Characteristic | Meaning |
|---|---|
| 2 range typical (normalized) | 35-100 (50-100) % |
| Measurement error SpO2 in the ranges: | |
|
±3% (±5%) |
|
±2% |
| Measurement range of pulse rate, typical (normalized) | 20-300 (30-300) bpm |
| Limits of permissible absolute error when measuring pulse rate in the range of 30 to 120 bpm | ±1 bpm |
| Limits of permissible relative error when measuring pulse rate in the range of 121 to 300 bpm | ±2 % |
| Pulse rate modes | Adult/neonatal |
| Visual indication of current states | “Missing sensor”, “Missing finger in sensor”, “Missing pulse”, battery status |
| Displaying parameters on the screen | SpO2, pulse rate, pulse beats, pulse wave (photoplethysmogram), pulse filling index, device status, spirogram |
| External monitoring software calculates and measures the following parameters | SpO2, duration of monitoring, duration of SpO2 analysis, total time of absence of pulse rate, duration of analysis of PR, SpO2min (%), SpO2max (%), SpO2avrg (%), PRmin (1/min), PRmax (1/min), PRavrg (1/min), PRage (1/min), PRmax/PRage (%), desaturation index, apnea/hypapnea index, perfusion index, BF, SpO2rest avrg – SpO2stress avg (%), SpO2rest avrg – SpO2stress min (%), recovery time SpO2, recovery time PR. |
| Data refresh rate | at least 1 time per second |
| Simultaneous registration of SpO2, pulse rate, photoplethysmogram, spirogram, body position indicator | within 24 hours |
| Dimensions | 122x80x24 mm |
| Weight (with batteries) | < 0.2 kg |
| Continuous operation time with one set of elements, min | 24 hours |
| Safety standards | STB IEC 60601-1-2-2006 STB EN 55011-2006 GOST 30324.0-95 refers to electrical products of class II, working part of BF (CF) type |